Safety: Pharmacodynamics in AURYXIA
AURYXIA has been shown to have an impact on serum phosphorus1
Mean serum phosphorus decreased from baseline in patients with AURYXIA1
AURYXIA (n=117)
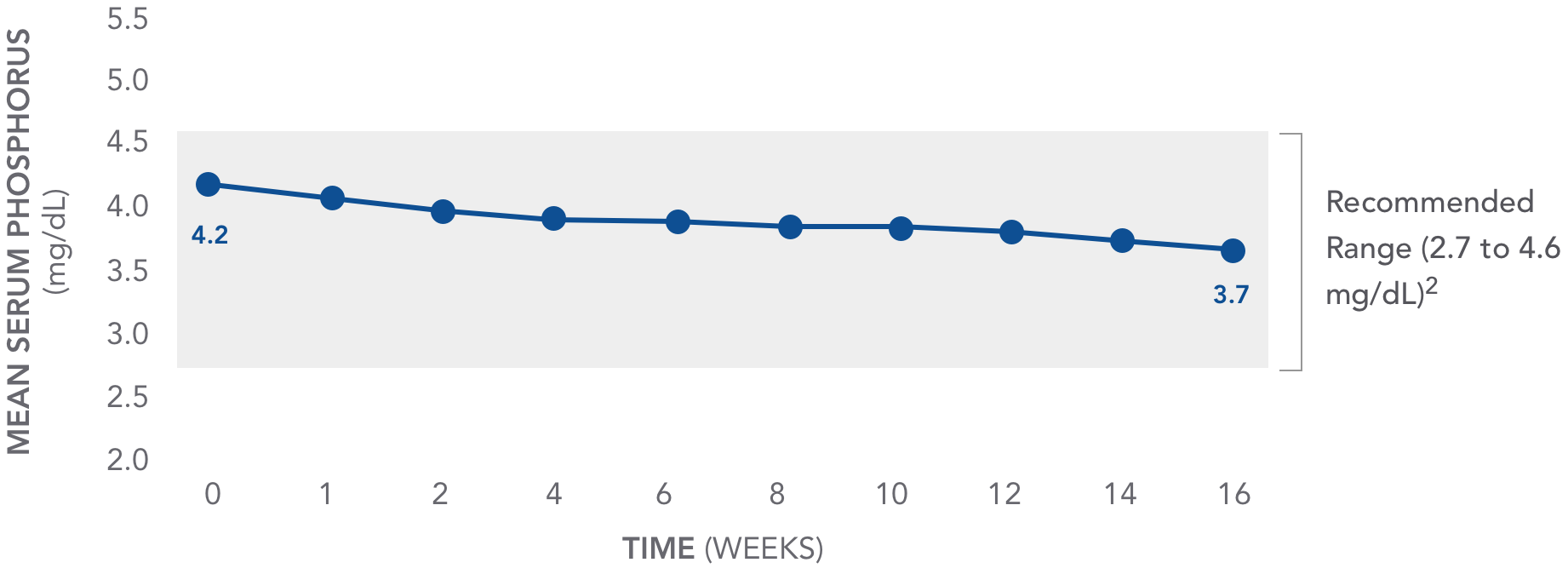
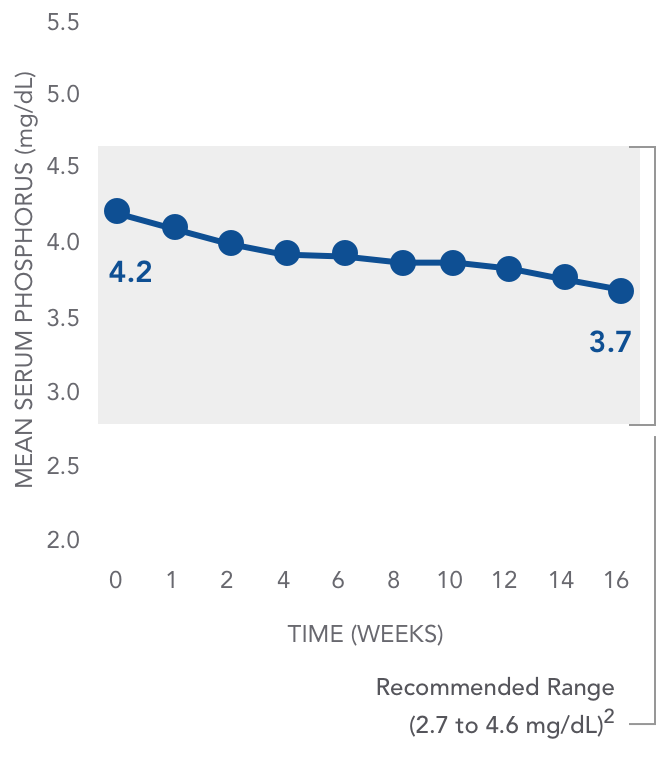
PHARMACODYNAMICS: PHOSPHORUS RANGE
- During the 16-week randomized period: Incidence of hypophosphatemia, reported as a treatment-related adverse event, occurred in 1% of patients treated with AURYXIA (1 patient) and 2% of patients treated with placebo (2 patients)3
You may also be interested in:
Trial design5
In a 24-week study consisting of a 16-week, randomized, double-blind, placebo-controlled efficacy period followed by an 8-week, open-label safety extension period, this trial evaluated the efficacy and safety of AURYXIA for the treatment of iron deficiency anemia in adult patients with CKD not on dialysis. Patients who were intolerant of or have had an inadequate therapeutic response to oral iron supplements, with hemoglobin ≥9.0 g/dL and ≤11.5 g/dL, serum ferritin ≤200 ng/mL, and TSAT ≤25% were enrolled. Patients were randomized to treatment with either AURYXIA (n=117) or placebo (n=117).
The primary endpoint was the proportion of patients achieving a ≥1.0 g/dL increase in hemoglobin at any time point during the 16-week efficacy period. Use of oral iron, IV iron, or ESAs was not permitted at any time during the trial.
CKD=chronic kidney disease; KDOQI=Kidney Disease Outcomes Quality Initiative.
