The first and only oral iron specifically indicated for adults with CKD-NDD
Discover the Hb and iron parameter results for AURYXIA from the 16-week pivotal trial.1-4
CKD=chronic kidney disease; NDD=non-dialysis dependent.
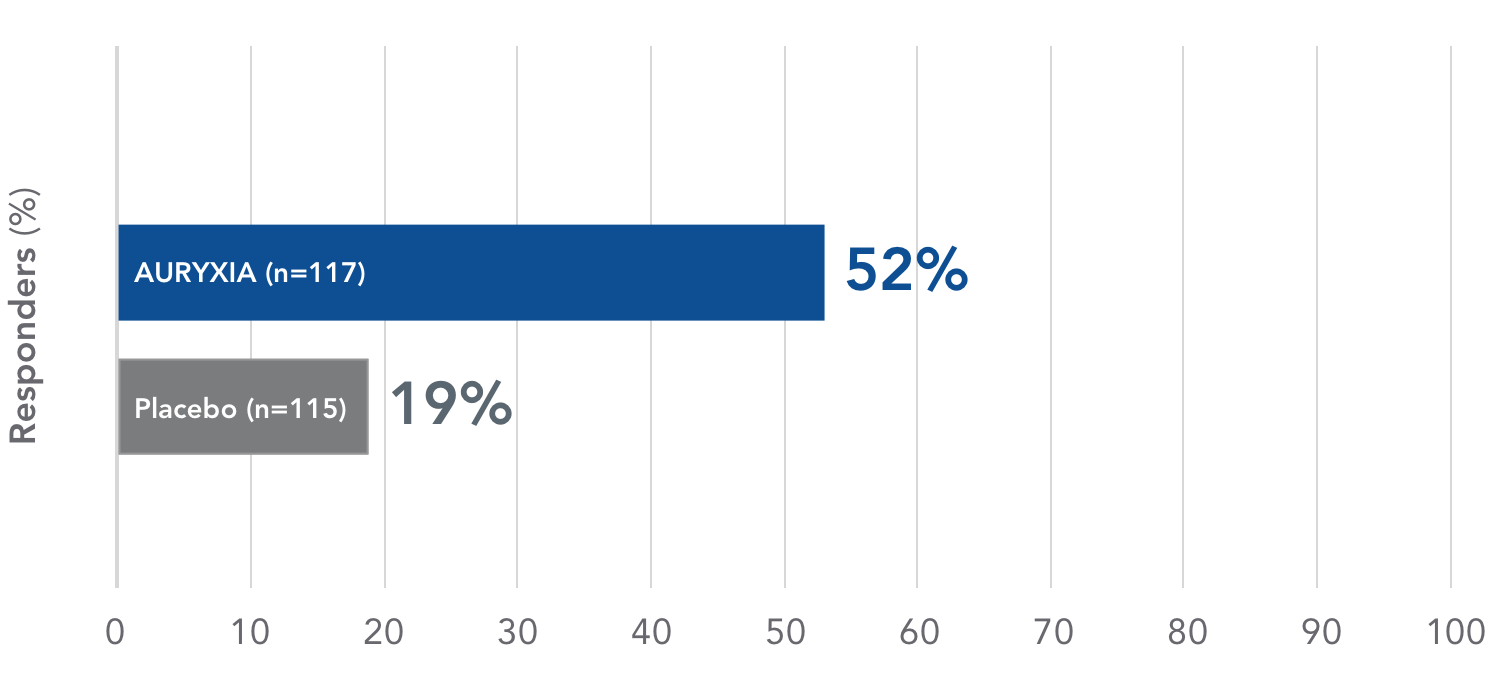
AURYXIA increased Hb ≥1.0 g/dL in the majority of treated patients1
Primary endpoint
Proportion of patients achieving an Hb increase of ≥1.0 g/dL at any time by Week 16
All efficacy endpoints were achieved without ESA or IV iron use, per exclusion criteria and study design.
VIEW DATA TABLE
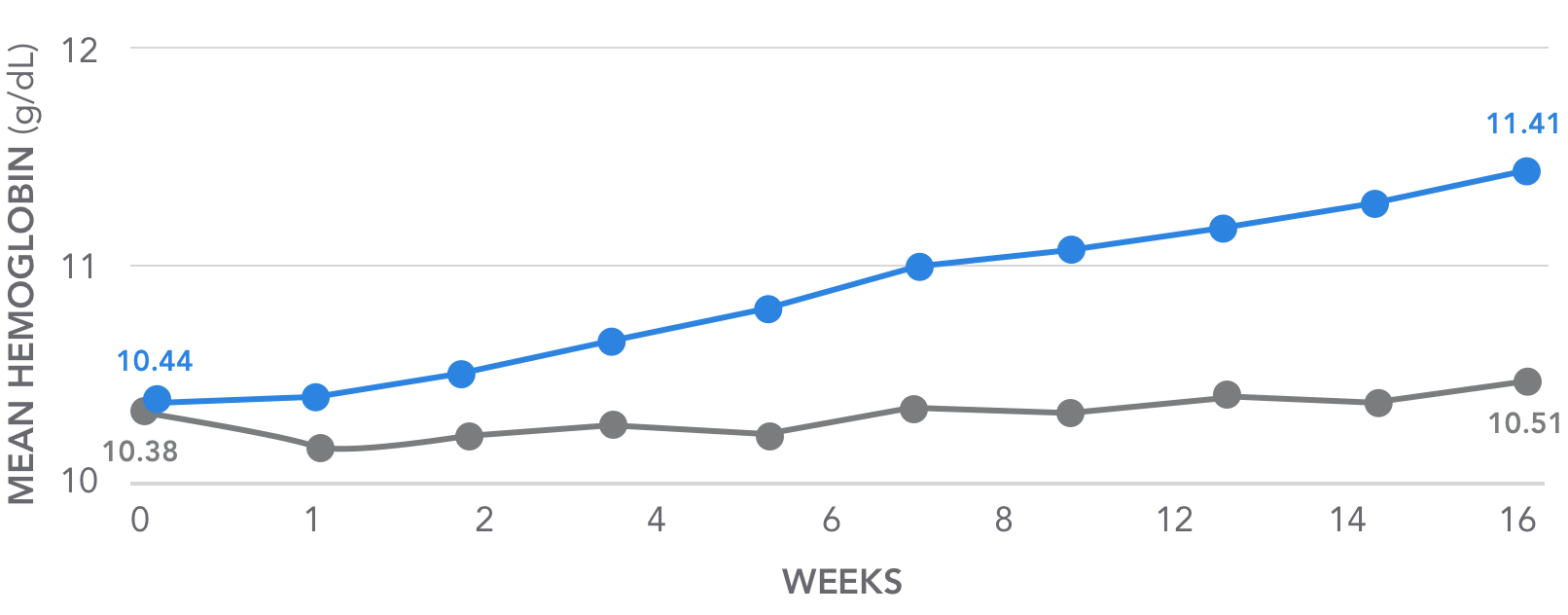
Summary of change in Hb (g/dL) from baseline over time during
randomized period based on observed values3
| FERRIC CITRATE | PLACEBO | ||
|---|---|---|---|
| Mean Hb (g/dL) | Mean Hb (g/dL) | Change from Baseline (P-Value) | |
| Baseline (n) | 10.44 (117) | 10.38 (115) | |
| Week 1 (n) | 10.45 (110) | 10.25 (107) | 0.11 (0.07) |
| Week 2 (n) | 10.55 (114) | 10.28 (103) | 0.24 (0.001) |
| Week 4 (n) | 10.68 (108) | 10.33 (100) | 0.33 (<0.001) |
| Week 6 (n) | 10.81 (102) | 10.29 (96) | 0.49 (<0.001) |
| Week 8 (n) | 10.98 (97) | 10.39 (91) | 0.57 (<0.001) |
| Week 10 (n) | 11.06 (97) | 10.37 (88) | 0.69 (<0.001) |
| Week 12 (n) | 11.16 (97) | 10.44 (85) | 0.69 (<0.001) |
| Week 14 (n) | 11.25 (97) | 10.42 (85) | 0.81 (<0.001) |
| Week 16 (n) | 11.41 (97) | 10.51 (77) | 0.84 (<0.001) |
AURYXIA increased mean hemoglobin levels over 16 weeks of treatment2,3
Proven efficacy in CKD-NDD: 52% of patients treated with AURYXIA achieved an Hb increase of ≥1.0 g/dL at any time point by Week 16 (P<0.001)1
Key secondary endpoint: Mean Hb increase from 10.4 g/dL to 11.4 g/dL by the end of the 16-week period2,3
AURYXIA (n=117)
PLACEBO (n=115)
- No IV iron or ESA use, as per trial design and exclusion criteria1
- Studied in patients who were unsuccessful with or intolerant of traditional oral iron therapy1
AURYXIA increased TSAT and ferritin over 16 weeks of treatment2
Key secondary endpoint: Mean change in TSAT over time4
AURYXIA (n=117)
PLACEBO (n=115)
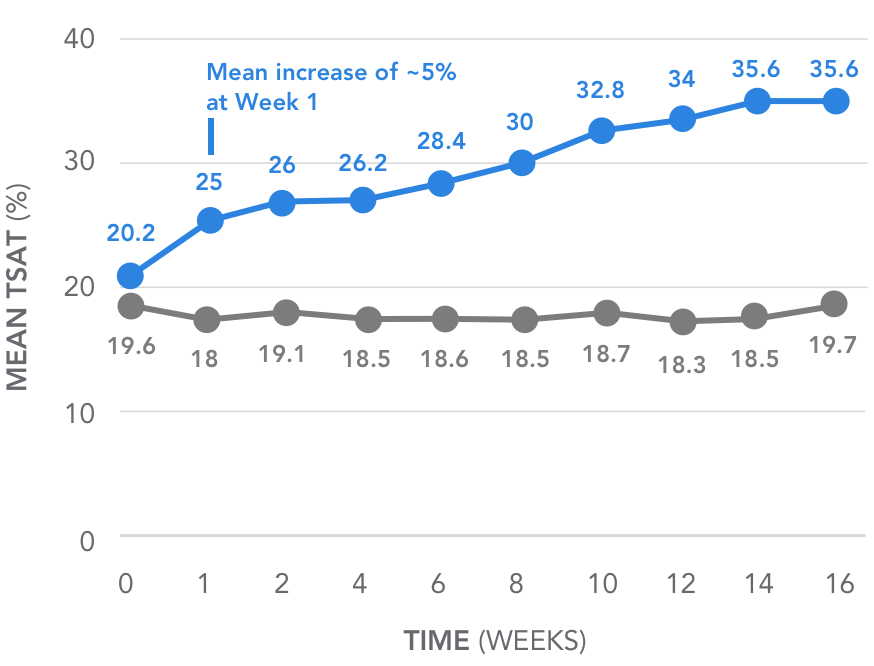
Over 16 weeks, AURYXIA increased mean TSAT from 20.2% to 35.6%4
(P<0.001)
Key secondary endpoint: Mean change in ferritin over time
AURYXIA (n=117)
PLACEBO (n=115)
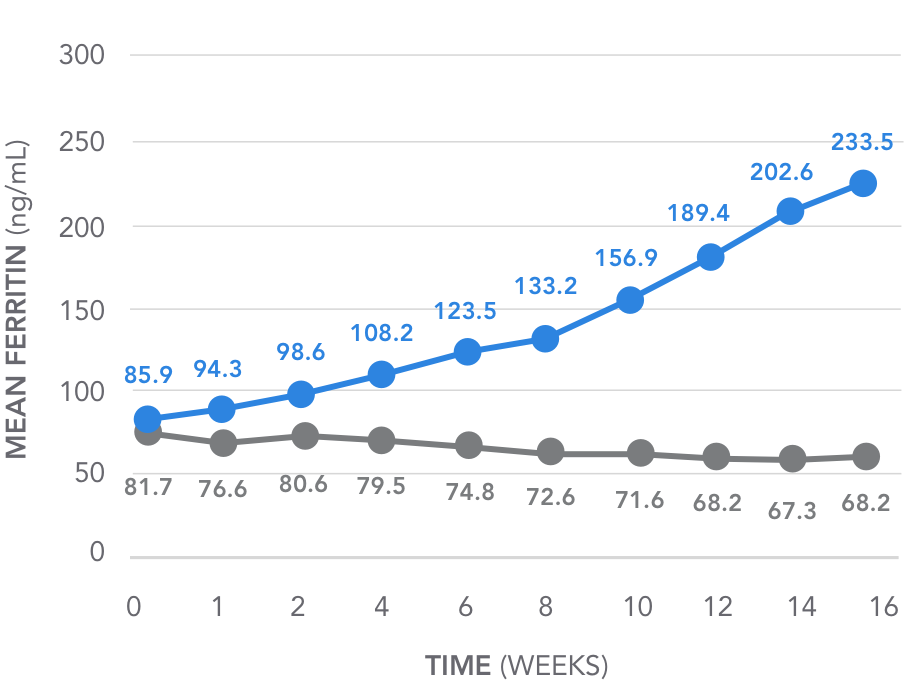
Over 16 weeks, AURYXIA increased mean ferritin from 85.9 ng/mL to 233.5 ng/mL5
(P<0.001)
(P<0.001)
(P<0.001)
- Patients on AURYXIA also achieved a mean increase in serum ferritin of 163 ng/mL from baseline (85.9 ng/mL) at Week 161,2
- No use of IV iron or ESAs, as per trial design and exclusion criteria1
- Studied in patients who were unsuccessful with or intolerant of traditional oral iron therapy1
Trial design1
In a 24-week study consisting of a 16-week, randomized, double-blind, placebo-controlled efficacy period followed by an 8-week, open-label safety extension period, this trial evaluated the efficacy and safety of AURYXIA for the treatment of iron deficiency anemia in adult patients with CKD not on dialysis. Patients who were intolerant of or have had an inadequate therapeutic response to oral iron supplements, with hemoglobin ≥9.0 g/dL and ≤11.5 g/dL, serum ferritin ≤200 ng/mL, and TSAT ≤25% were enrolled. Patients were randomized to treatment with either AURYXIA (n=117) or placebo (n=117).
The primary endpoint was the proportion of patients achieving a ≥1.0 g/dL increase in hemoglobin at any time point during the 16-week efficacy period. Use of oral iron, IV iron, or ESAs was not permitted at any time during the trial.
Key secondary endpoints included:
- Mean changes in hemoglobin, TSAT, ferritin, and phosphorus from baseline to Week 16
- Proportion of patients experiencing a sustained treatment effect on hemoglobin (≥0.75 g/dL mean change from baseline over any 4-week period provided that an increase of at least 1.0 g/dL had occurred during that 4-week period)
CKD=chronic kidney disease; Hb=hemoglobin; IV=intravenous; ESAs=erythropoiesis-stimulating agents; TSAT=transferrin saturation.
